Global landscape analysis of no-fault compensation programmes for vaccine injuries: A review and survey of implementing countries | PLOS ONE
Health Resources and Services Administration Department of Health and Human Services Attention: HRSA Regulations Officer, 5600 F

Jurisdictions with Vaccine Injury Compensation Programs (including the... | Download Scientific Diagram

The National Vaccine Injury Compensation Program and the Office of Special Masters - EveryCRSReport.com
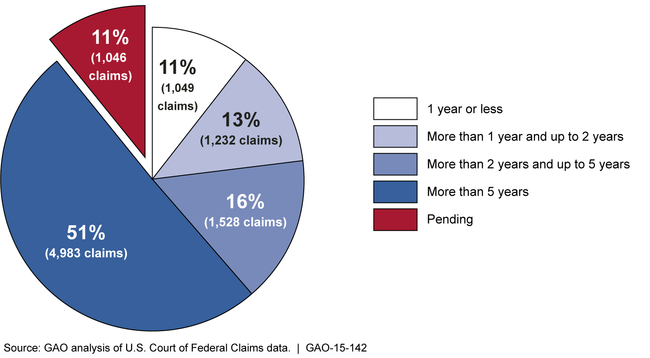
Vaccine Injury Compensation: Most Claims Took Multiple Years and Many Were Settled through Negotiation | U.S. GAO
Vaccine Injury Compensation: Program Challenged to Settle Claims Quickly and Easily - Page 8 of 48 - UNT Digital Library











