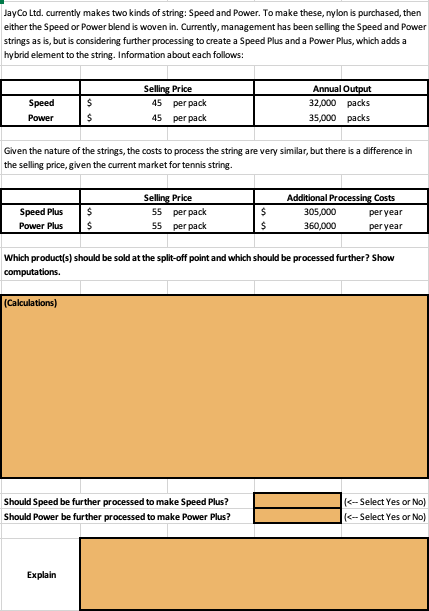
A compound is formed by two elements M and N. The element N forms ccp and atoms of M occupy 1/3rd of tetrahedral voids. What is the formula of the compound?

If two elements can combine to form more than one compound, the masses of one element that combine with a fixed mass of the other element, are in whole number ratio. (a)