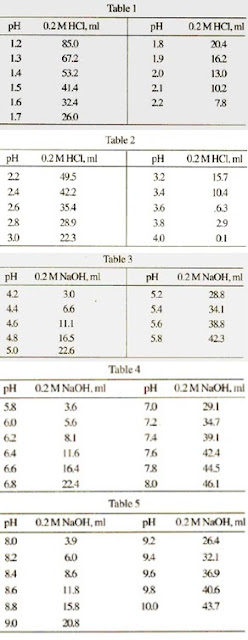
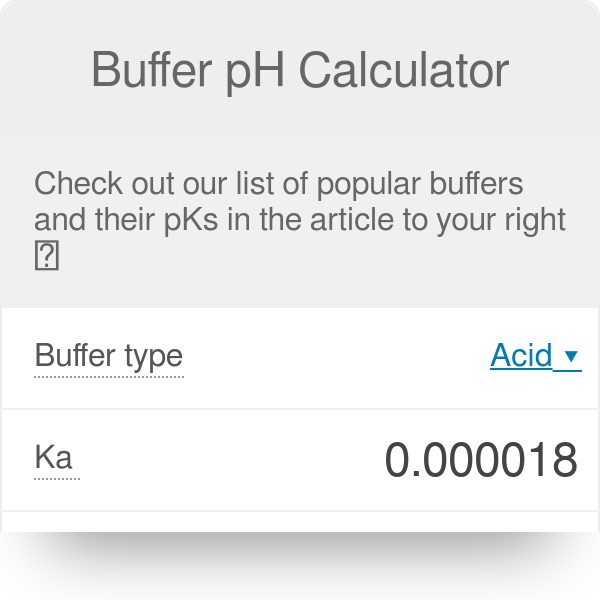
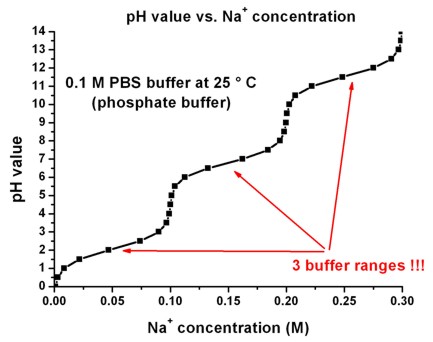
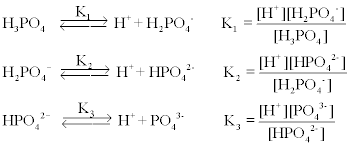The pKa of the phosphate buffer system is 7.2. Using the Henderson Hasselbach equation, what is the final pH of the environment after adding 25 mL of 0.01 M HCl to 1

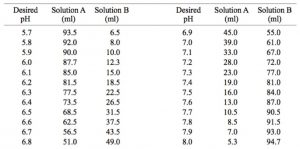
pH calculations and more in fundamentals of pharmaceutics. : Prepare 1 liter phosphate buffer solution at pH = 7.4 and with a buffer capacity of 0.1