Optimised Conditions for the Palladium-Catalyzed Hydrogenolysis of Benzyl and Naphthylmethyl Ethers: Preventing Saturation of Aromatic Protecting Groups | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Palladium-catalysed carboformylation of alkynes using acid chlorides as a dual carbon monoxide and carbon source | Nature Chemistry

Palladium‐Catalysed Decarboxylative Generation and Regiodivergent Prenylation of 2‐Azaallyl Anions - Wang - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library
Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands

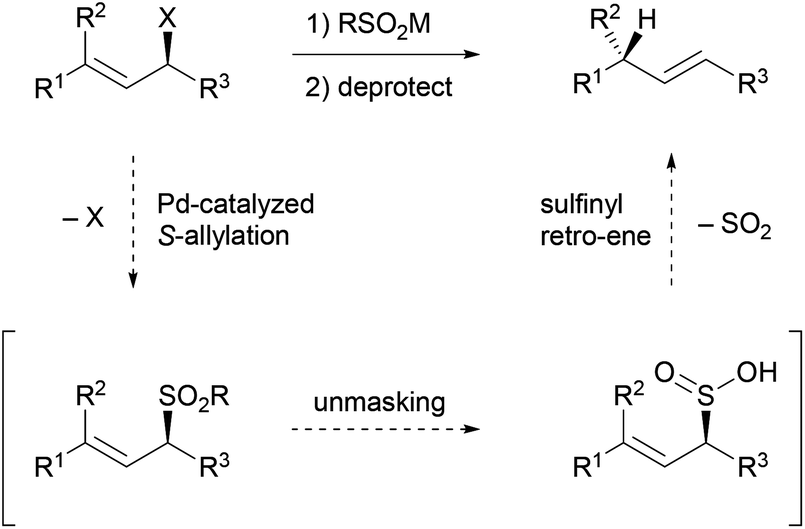
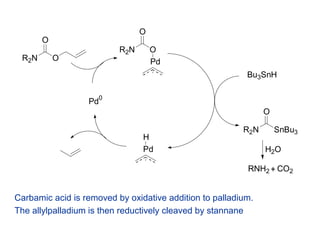
Mechanism of allyl deprotection through catalytic palladium π-allyl... | Download High-Resolution Scientific Diagram
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions

Propargylic and Allenic Carbocycle Synthesis through Palladium-Catalyzed Dearomatization Reaction | The Journal of Organic Chemistry

Safe Removal of the Allyl Protecting Groups of Allyl Esters using a Recyclable, Low‐Leaching and Ligand‐Free Palladium Nanoparticle Catalyst - Takagi - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library

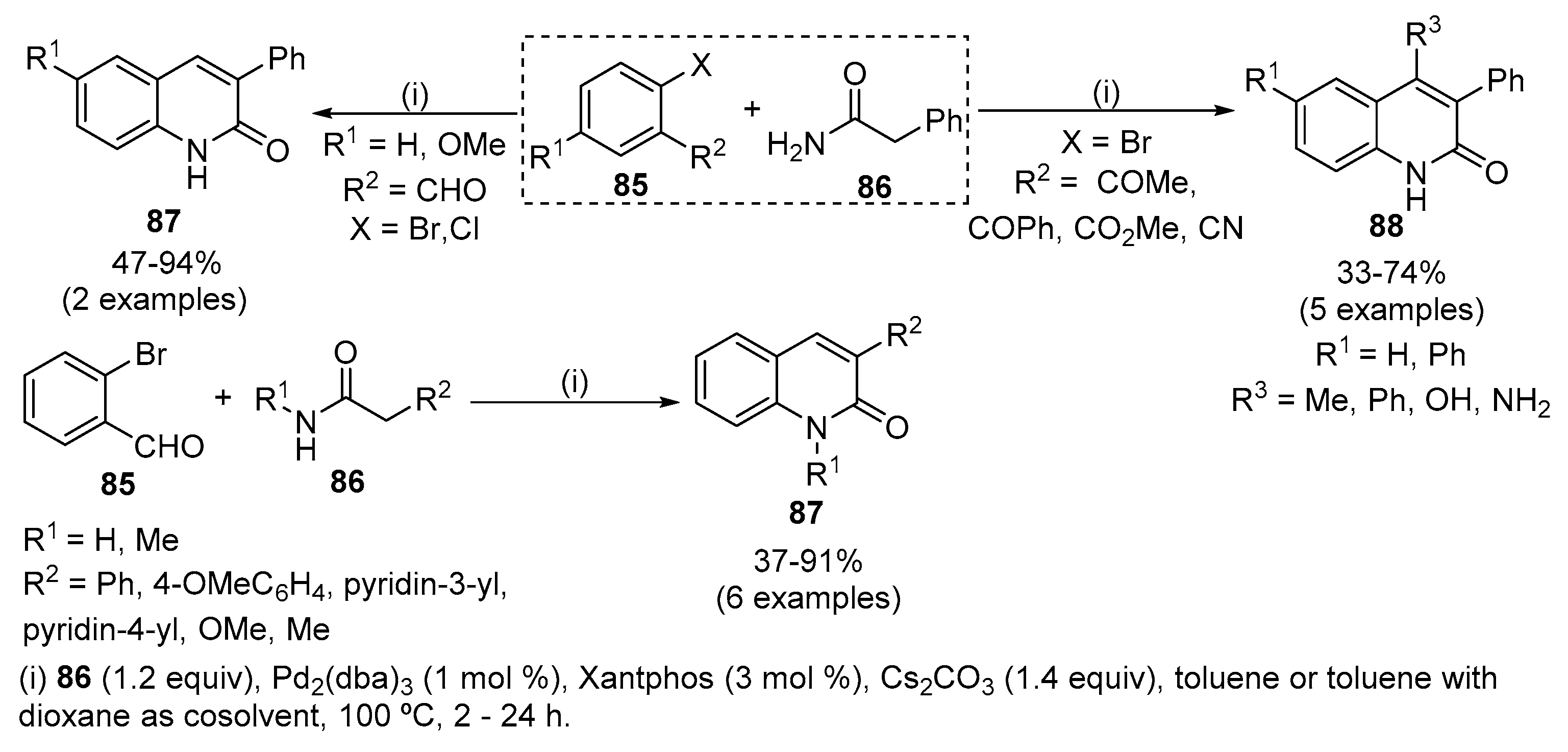
Scope of palladium-catalysed decarboxylative alkylation of lactams.a,... | Download Scientific Diagram

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands | ACS Catalysis
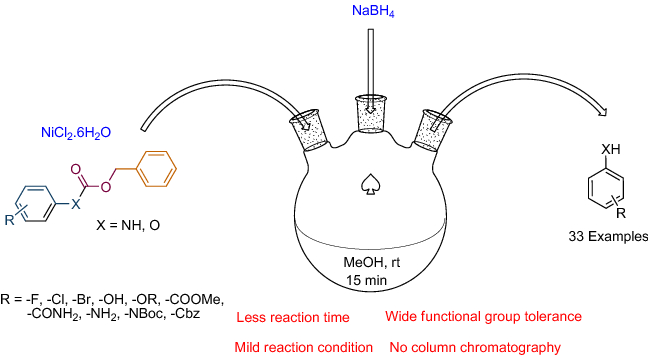
Development of a novel protocol for chemoselective deprotection of N/O-benzyloxycarbonyl (Cbz) at ambient temperature | SpringerLink
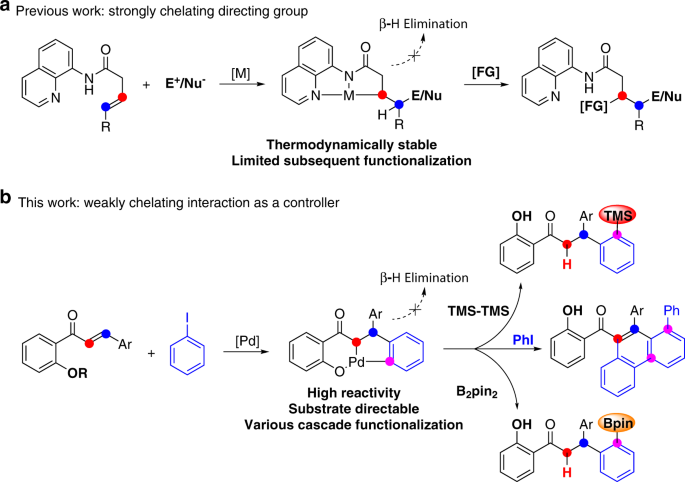
Quadruple C-H activation coupled to hydrofunctionalization and C-H silylation/borylation enabled by weakly coordinated palladium catalyst | Nature Communications