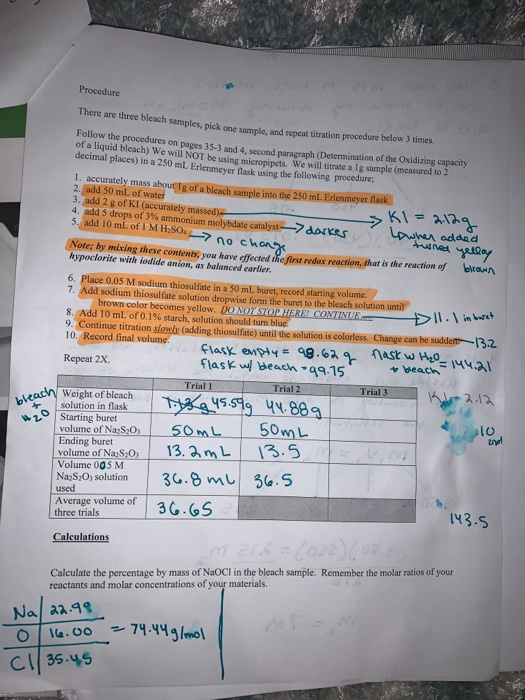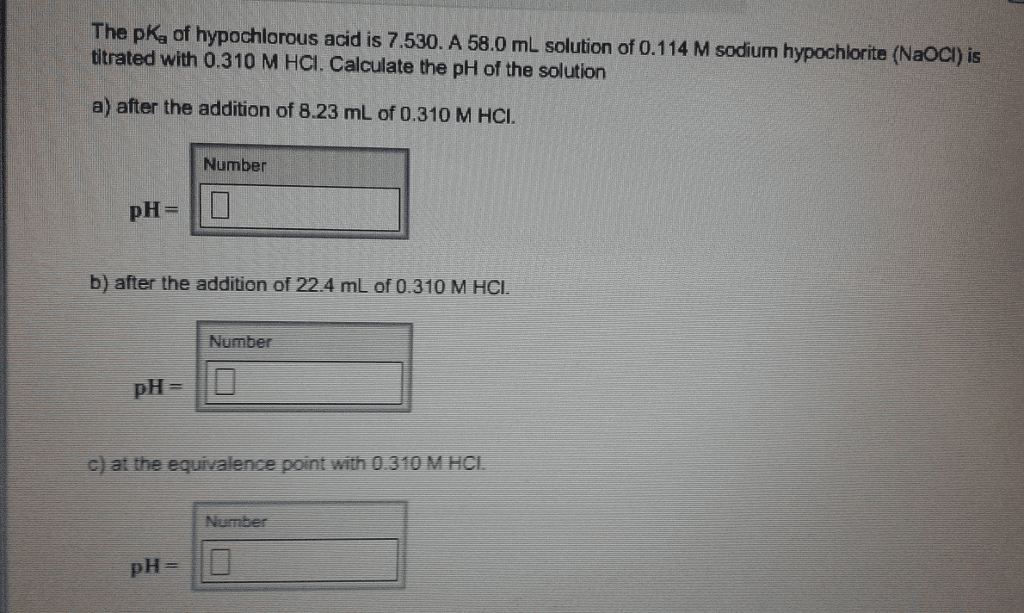
OneClass: The pKa of hypochlorous acid is 7.530. A 58.0 mL solution of 0.114 M sodium hypochlorite (N...
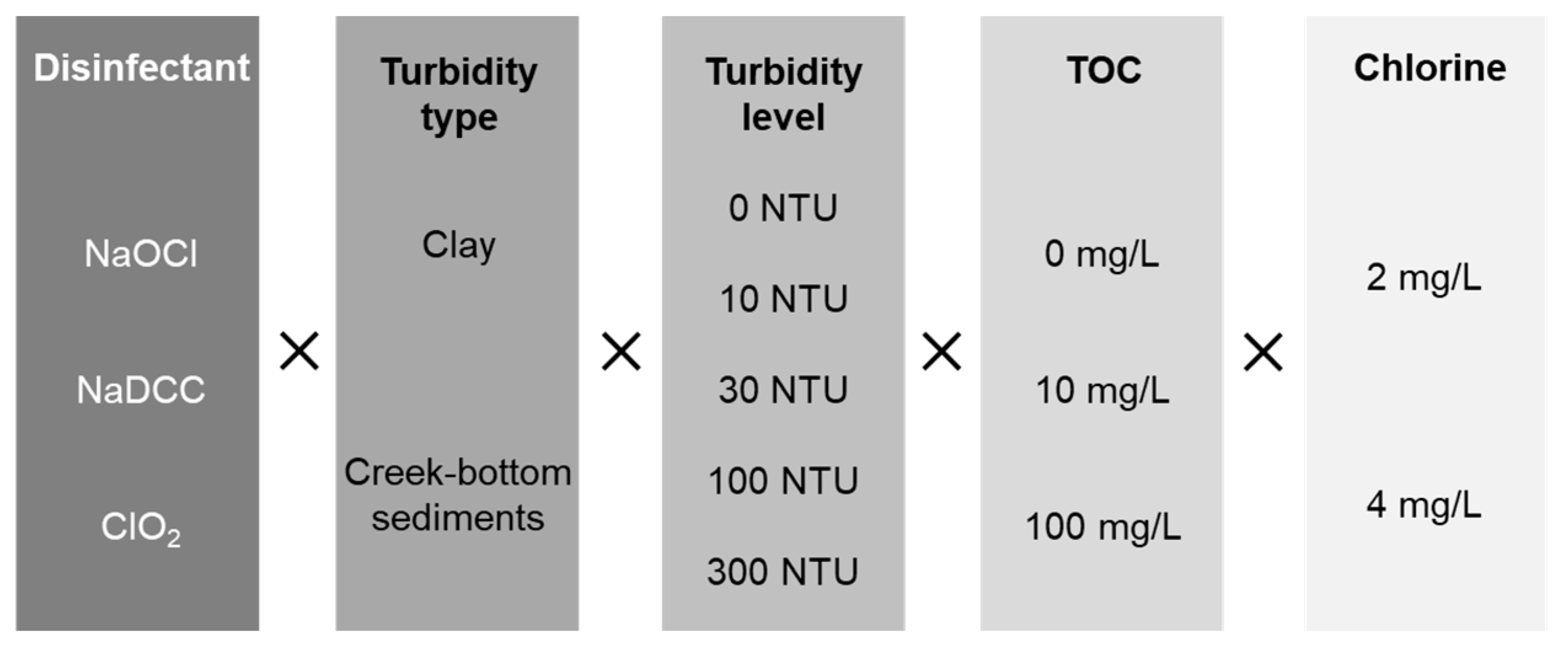
Water | Free Full-Text | Residual Maintenance Using Sodium Hypochlorite, Sodium Dichloroisocyanurate, and Chlorine Dioxide in Laboratory Waters of Varying Turbidity | HTML

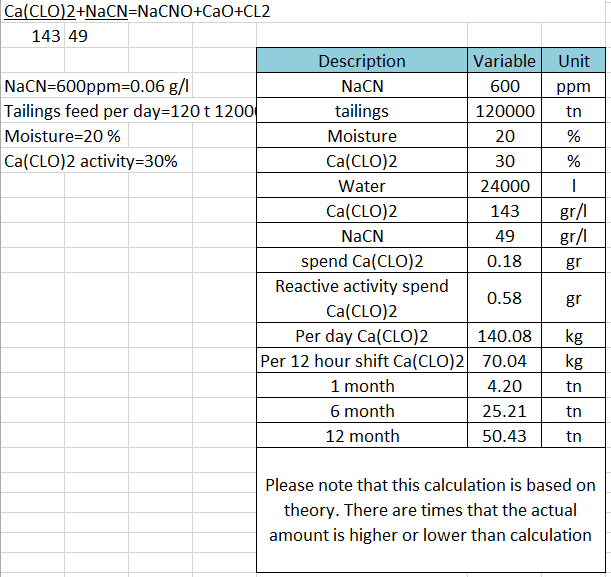
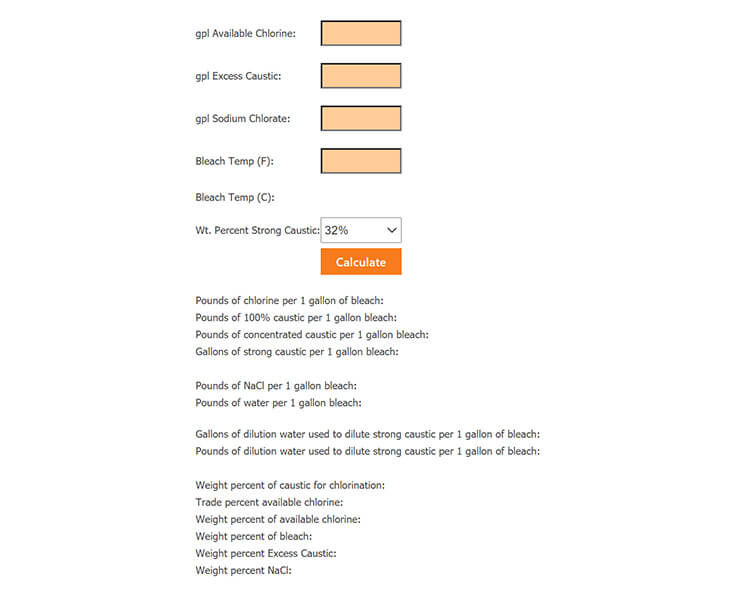
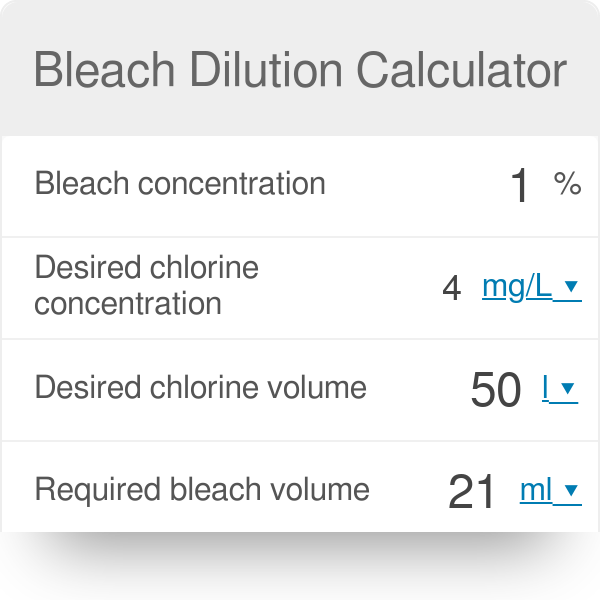
How do you calculate the concentration of sodium hypochlorite solution from the available chlorine percentage? | ResearchGate
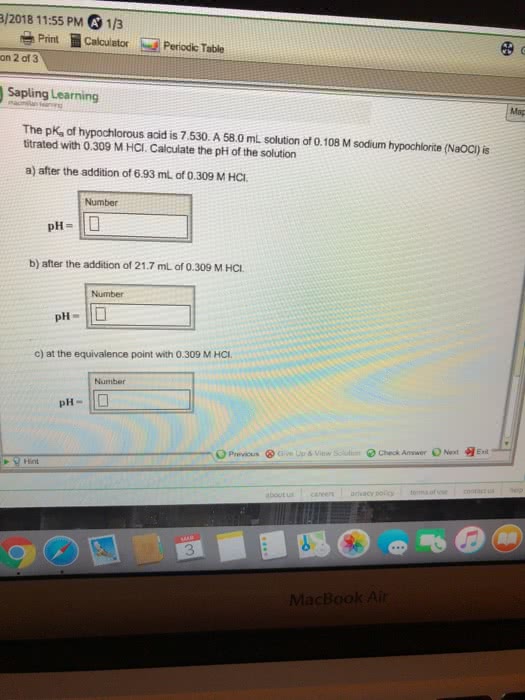
OneClass: The pKa of hypochlorous acid is 7.530. A 56.0 mL solution of 0.145 M sodium hypochlorite (N...
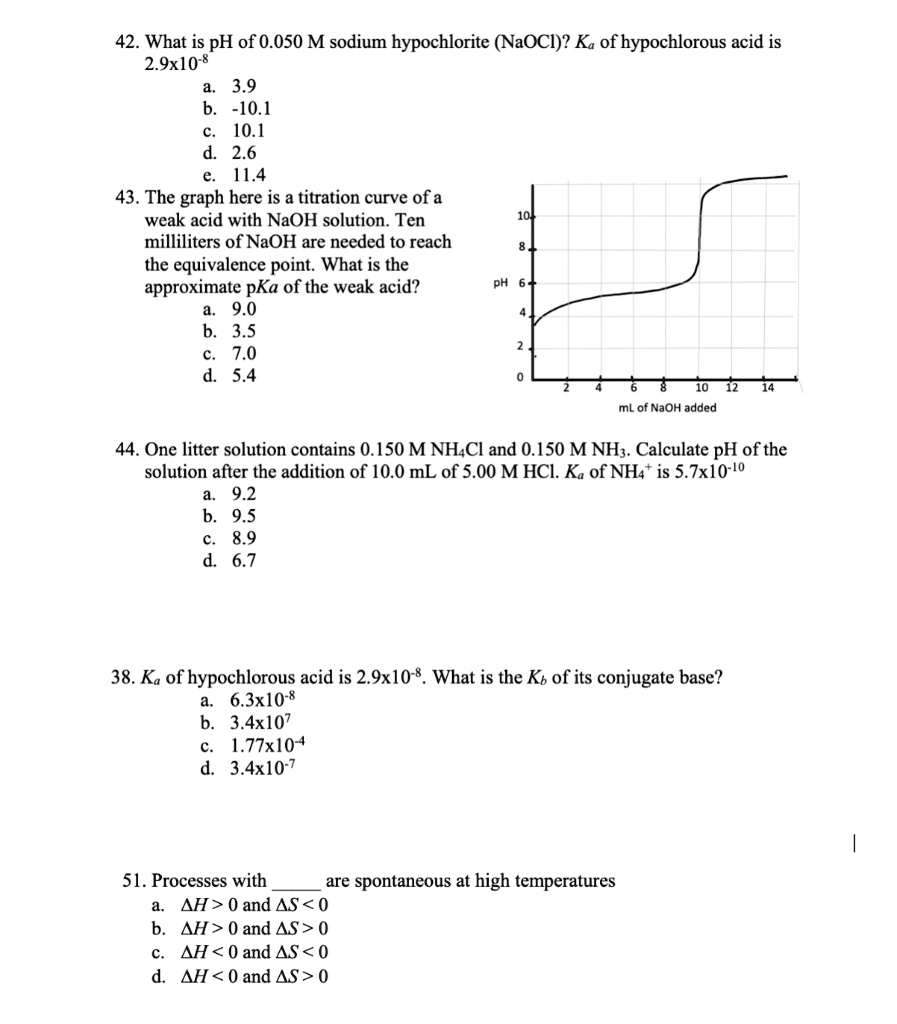
SOLVED: 42. What is pH of 0.050 M sodium hypochlorite (NaOCl)? Ka ofhypochlorous acid is 2.9xl0 8 3.9 -10.1 10.1 2.6 11.4 43. The graph here is a titration curve of a

How do you calculate the concentration of sodium hypochlorite solution from the available chlorine percentage? | ResearchGate