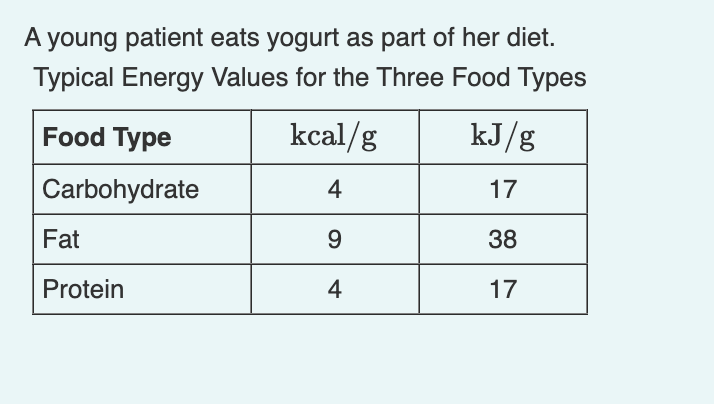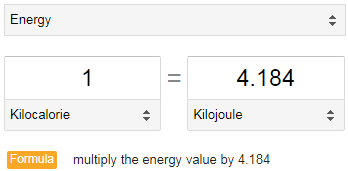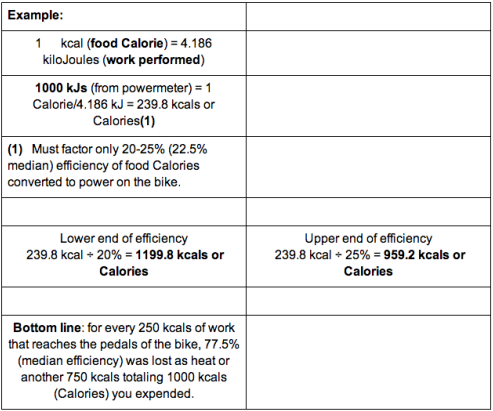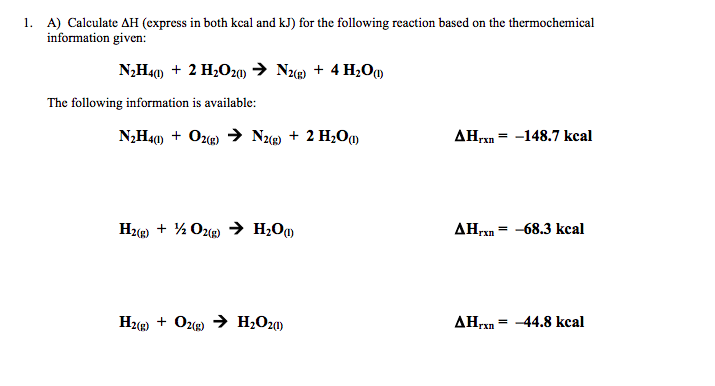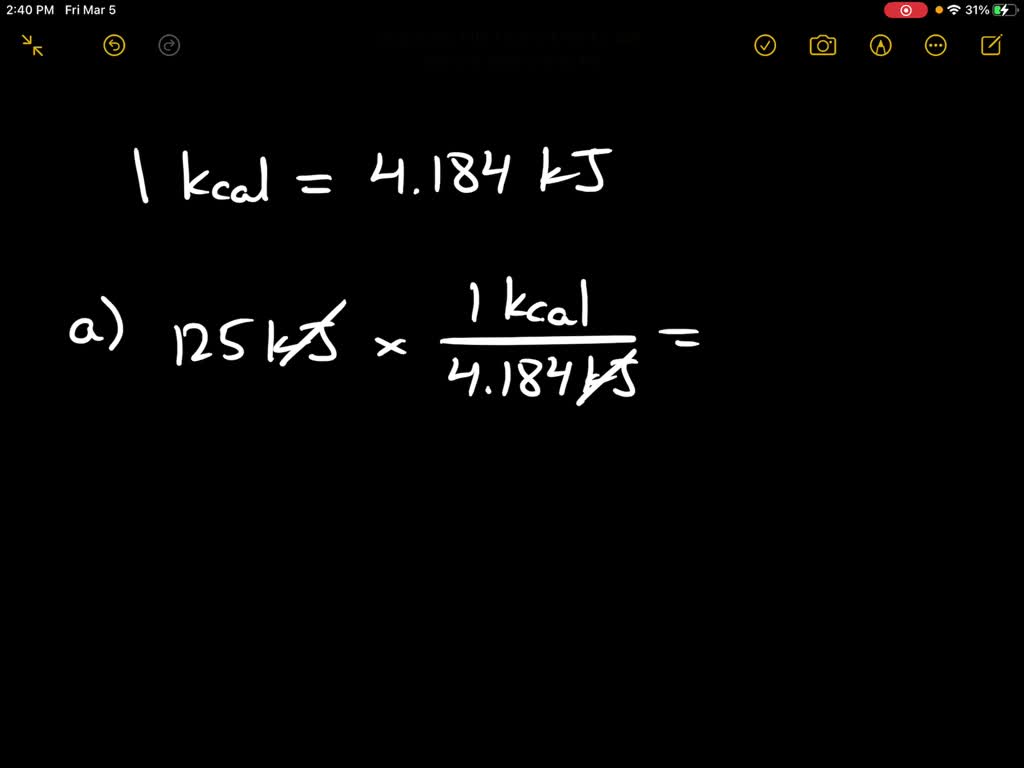
SOLVED:Calculate the kilocalories for each of the following: a. one stalk of celery that produces 125 kJ when burned in a calorimeter b. a waffle that produces 870 . k J when

SOLVED:Perform the indicated conversions. a. 91.74 kcal into calories b. 1.781 kJ into calories c. 4.318 ×10^3 J into kilocalories d. 9.173 ×10^4 cal into kilojoules