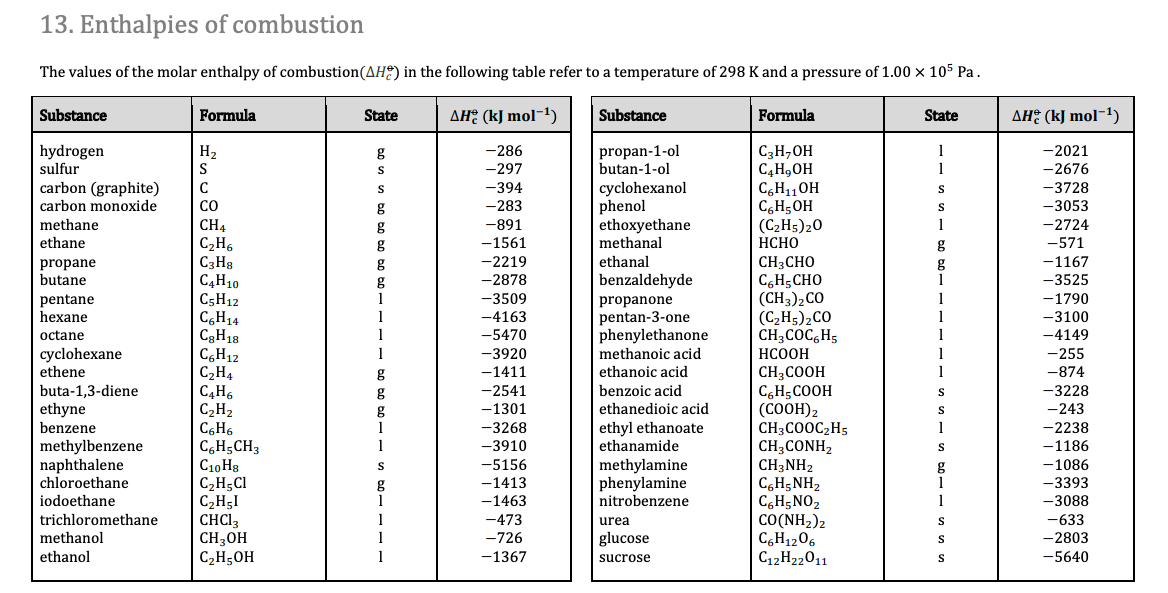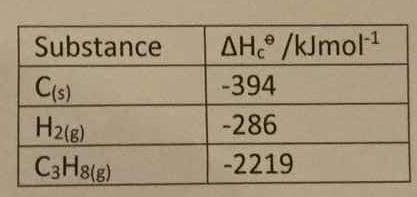
How do you determine the enthalpy change for the reaction below using the enthalpy of combustion data in the table: 3C_((s)) +4H_(2(s)) -> C_3H_(8(s))? | Socratic

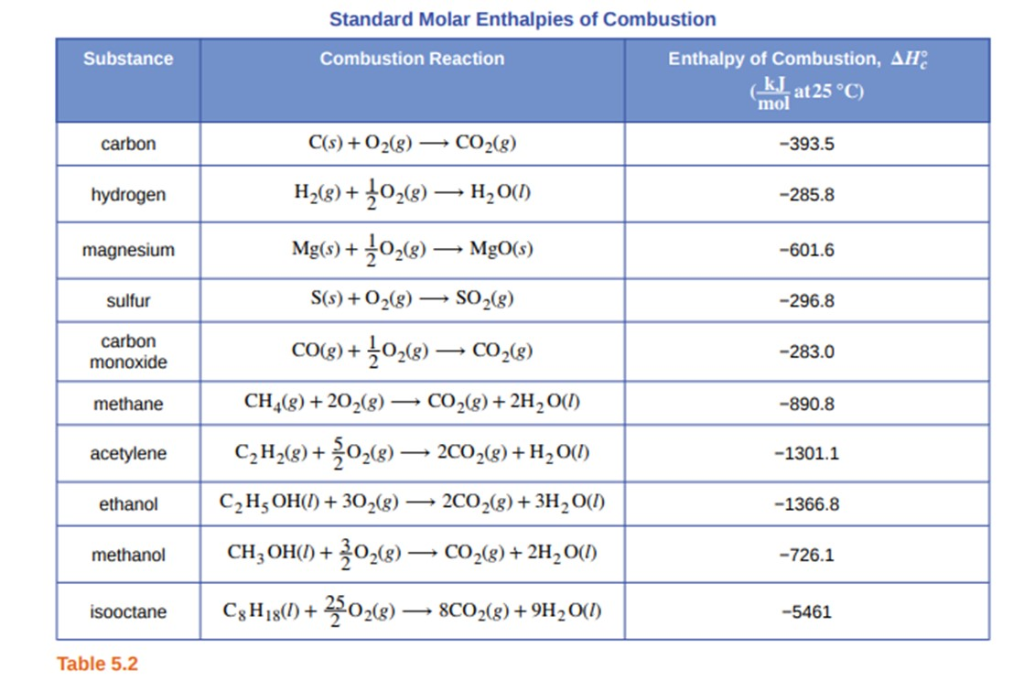
SOLVED: Standardl Molar Enthalpies ol Combustion Enthalpy ol Combustion AH? < (kJfmol at 258 C) 393.5 285.8 Substance Combustion Reaction Carbon (s) + 02 (g C0z Hydrogen [y 102 HzO Magnesium Mg
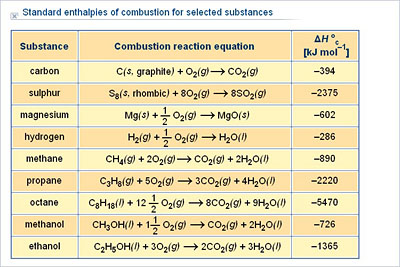
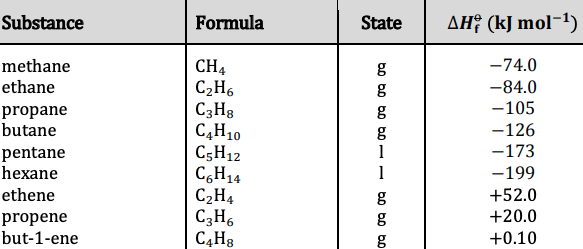
Chemistry - Upper Secondary - YDP - Chart - Standard enthalpies of combustion for selected substances

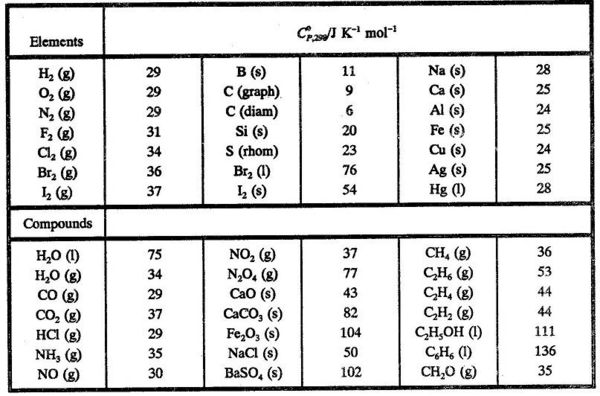
Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa