120ml/bottle Ema Nail Art Acrylic Liquid Monomer Polish Polymer For Polish Crystal Acrylic Powder Carving/dipping/extension Kt#4 - Acrylic Powders & Liquids - AliExpress

The Alstom/Cosider consortium and EMA celebrate the entry into commercial service of part 1 of the Constantine tramway line extension | Alstom
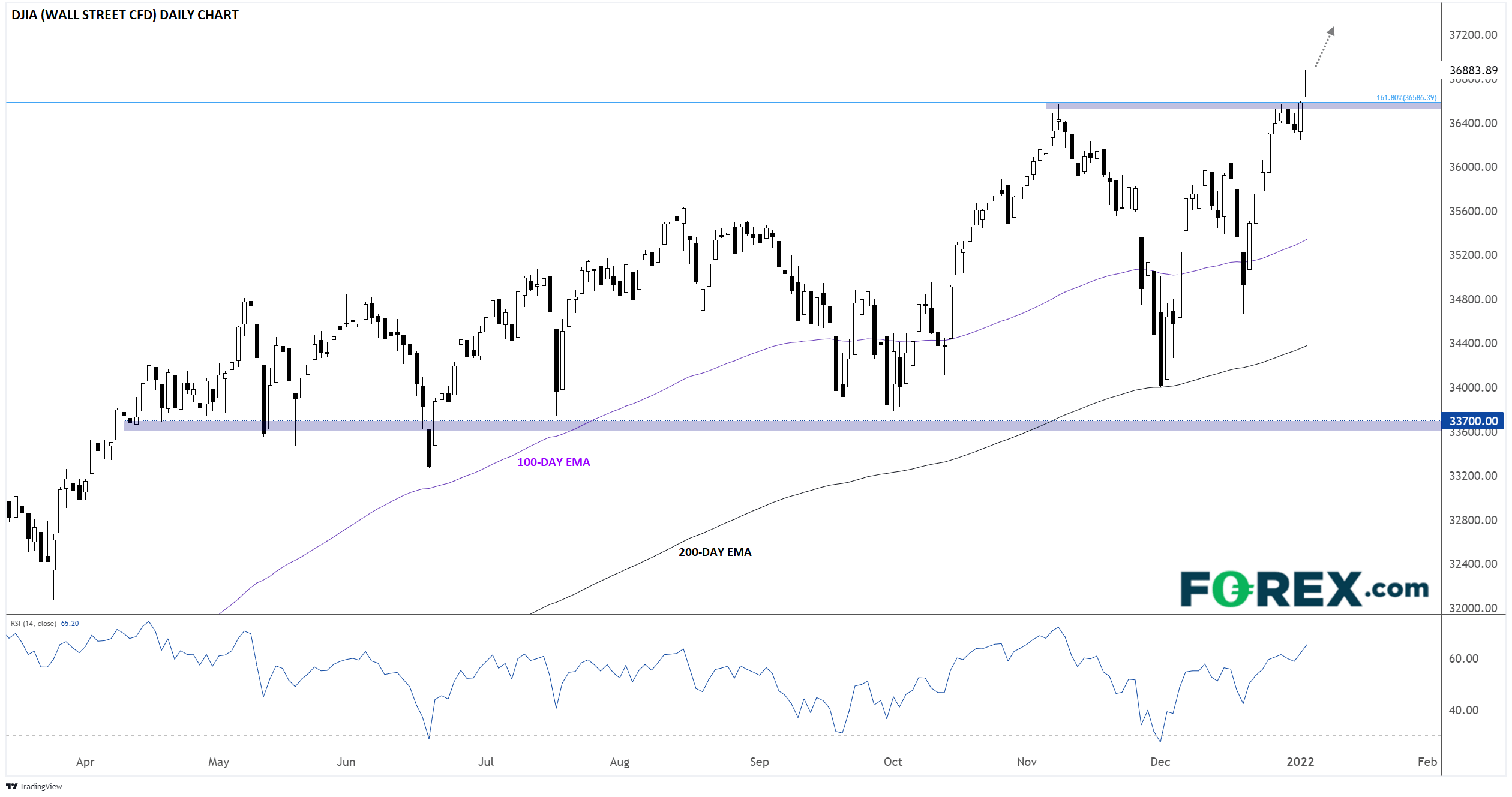
The impact of FDA and EMA regulatory decision-making process on the access to CFTR modulators for the treatment of cystic fibrosis | Orphanet Journal of Rare Diseases | Full Text

Buy Modelones Monomer Acrylic Nail Liquid -EMA Monomer- 4 oz Professional Nail Monomer Liquid, MMA-Free Acrylic Liquid for Acrylic Powder Acrylic Nail Art Nail Extension, Medium Drying, Non-Yellow Online at Lowest Price






:max_bytes(150000):strip_icc()/dotdash_Final_Strategies_Applications_Behind_The_50_Day_EMA_INTC_AAPL_Jul_2020-01-0c5fd4e9cb8b49ec9f48cb37d116adfd.jpg)