18.67 Consider the reaction Given that DG8 for the reaction at 258C is 173.4 kJ/mol, (a) calculate - Brainly.com
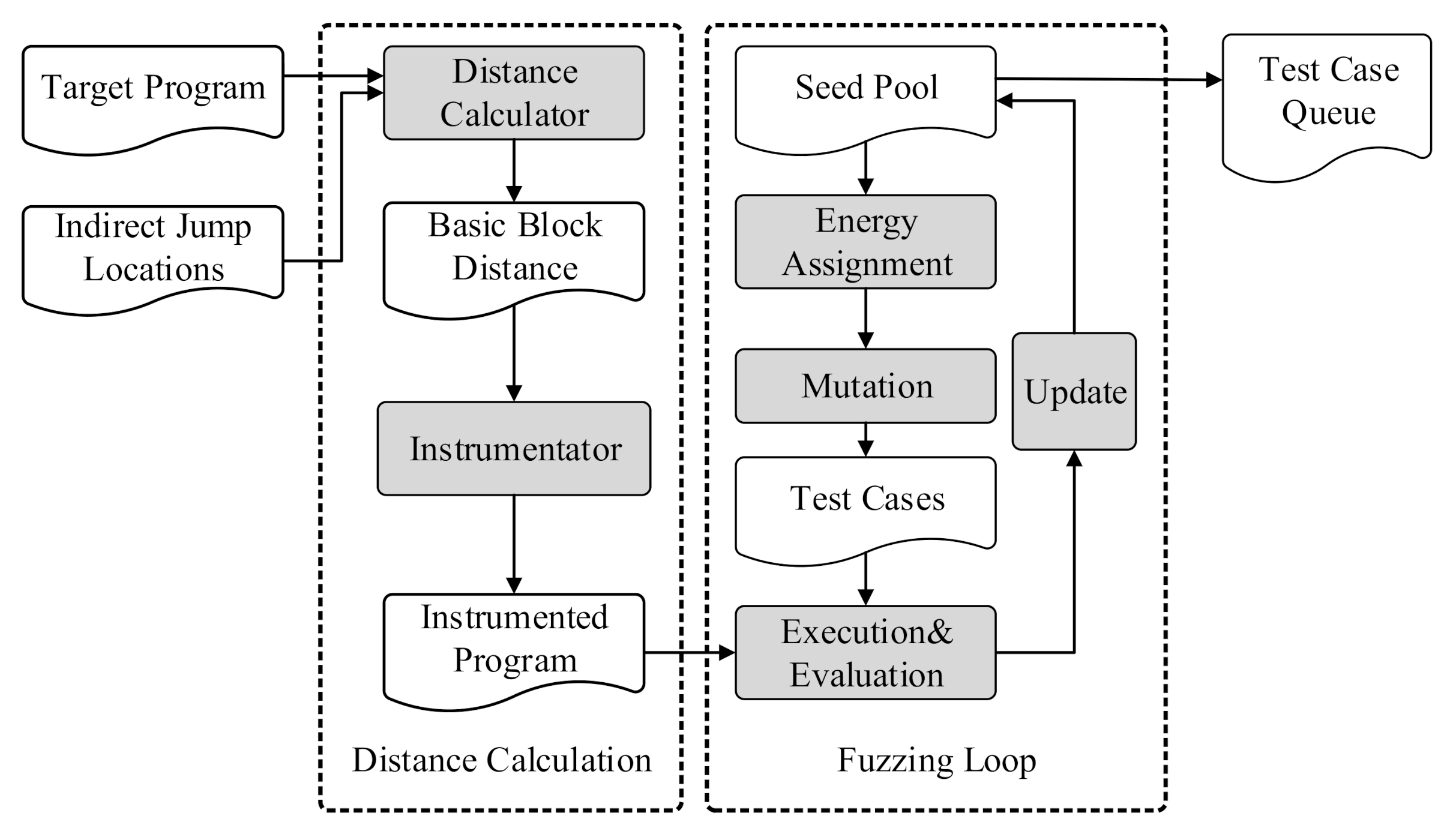
Applied Sciences | Free Full-Text | Constructing More Complete Control Flow Graphs Utilizing Directed Gray-Box Fuzzing | HTML
Energy Vault to provide 1.6 GWh of gravity energy storage to support DG Fuels SAF projects - Green Car Congress

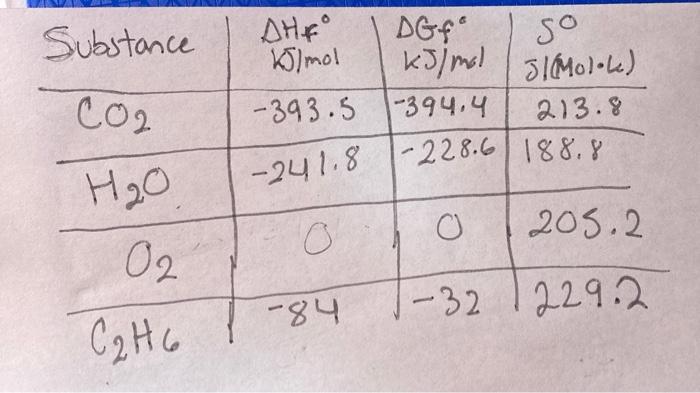
SOLVED: Calculate ΔG ̊ for the following reaction, given the ΔG˚f values of the substances in the table below. 2 Sb (s) + 6 HCl (g) → 2 SbCl3 (l) + 3

Optimization of wind/solar energy microgrid by division algorithm considering human health and environmental impacts for power-water cogeneration - ScienceDirect