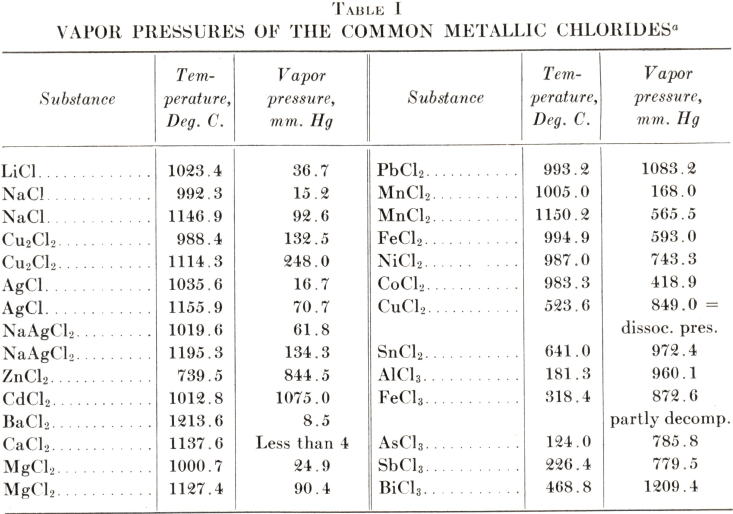
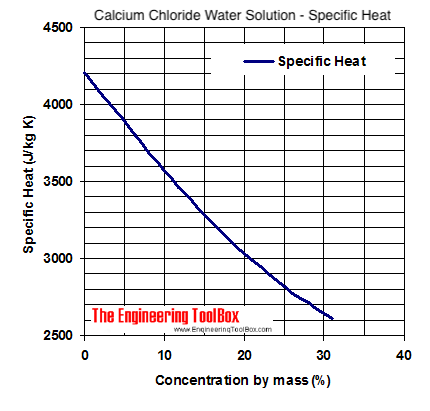
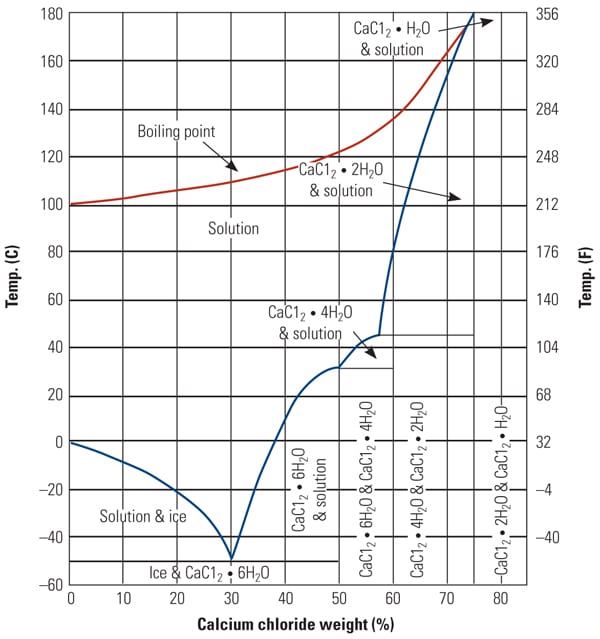
Measuring the Changes Due To Addition of Calcium Chloride in Acetic Acid Water Mixture and Generate the VLE Data with the Help o

Vapour pressure (in mbar) over pure water/ice and eutectic (CaCl 2 -H 2... | Download High-Quality Scientific Diagram

The vapour pressure of water over saturated solutions of sodium sulfate, calcium bromide, ferric chloride, zinc nitrate, calcium nitrate, and lithium nitrate at temperatures from 278.15 K to 323.15 K - ScienceDirect

SOLVED:Which of the following aqueous solutions has (a) the higher boiling point, (b) the higher freezing point, and (c) the lower vapor pressure: 0.35 m CaCl2 or 0.90 m urea? Explain. Assume

Measurements of Vapor Pressures of Aqueous Solutions in the NaCl–KCl–H2O System from 493.15 to 693.25 K in a Fused Silica Capillary High-Pressure Optical Cell | Journal of Chemical & Engineering Data
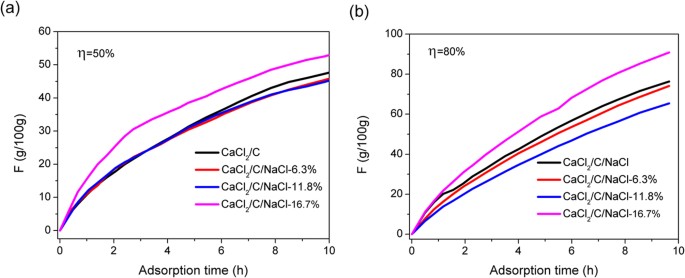
Steady-state equation of water vapor sorption for CaCl2-based chemical sorbents and its application | Scientific Reports

Vapor pressure of magnesium in the reduction of magnesium compounds... | Download Scientific Diagram

Vapor pressures of H 2 O and HCl over the magnesium chloride hydrates... | Download Scientific Diagram
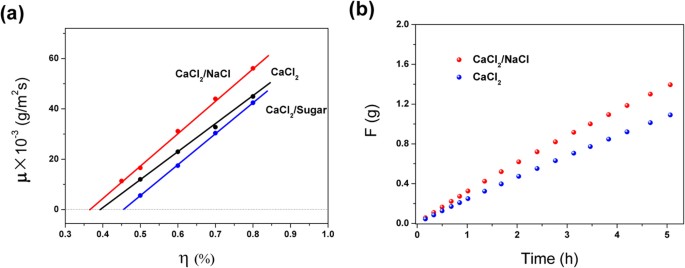
Steady-state equation of water vapor sorption for CaCl2-based chemical sorbents and its application | Scientific Reports
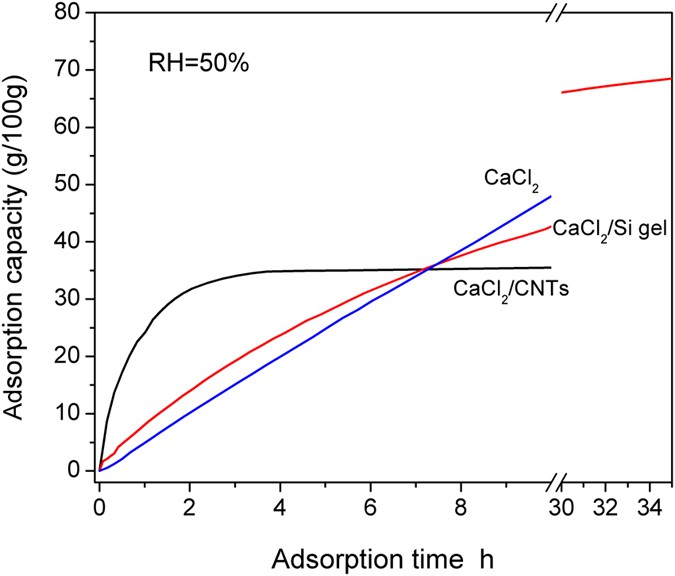
Steady-state equation of water vapor sorption for CaCl2-based chemical sorbents and its application | Scientific Reports
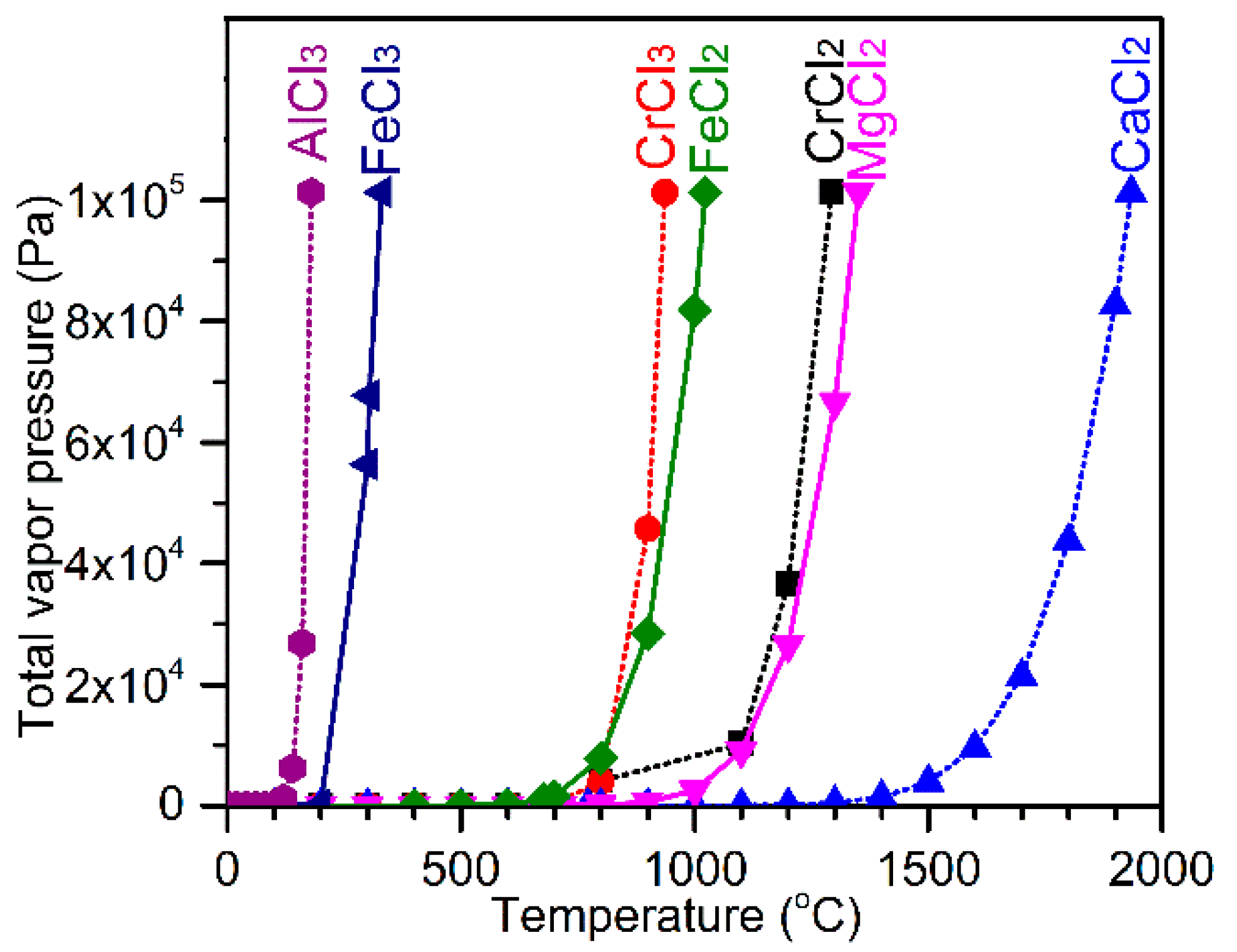
Minerals | Free Full-Text | Calcium Chloride-Assisted Segregation Reduction of Chromite: Influence of Reductant Type and the Mechanism | HTML

Equilibrium vapor pressure over aqueous solutions of NaCl (calculated... | Download Scientific Diagram