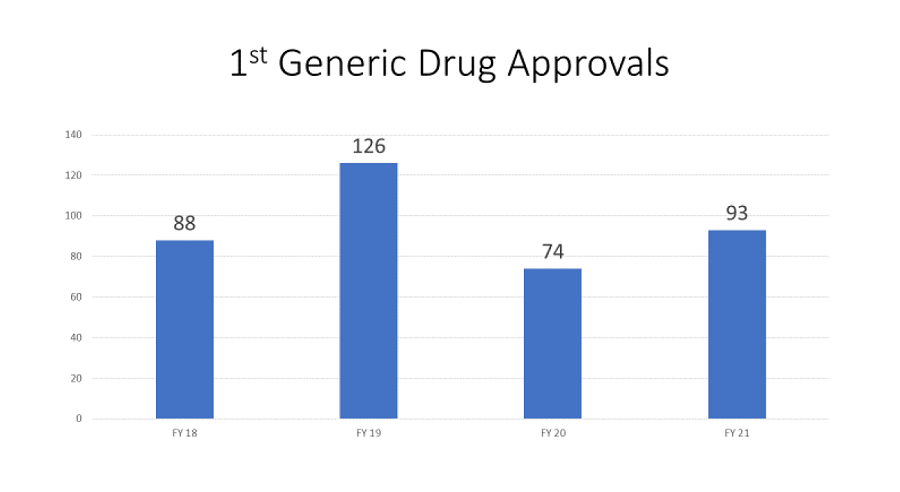
Association between US Pharmacopeia (USP) monograph standards, generic entry and prescription drug costs | PLOS ONE
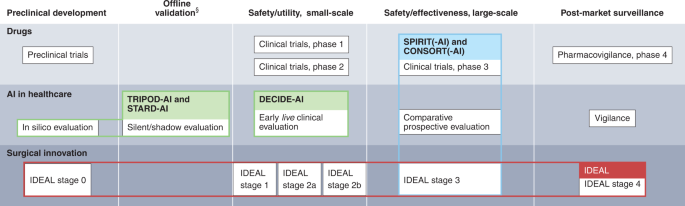
Reporting guideline for the early-stage clinical evaluation of decision support systems driven by artificial intelligence: DECIDE-AI | Nature Medicine

Metabolism by Aldehyde Oxidase: Drug Design and Complementary Approaches to Challenges in Drug Discovery | Journal of Medicinal Chemistry








![PDF] CRISP Data Mining Methodology Extension for Medical Domain | Semantic Scholar PDF] CRISP Data Mining Methodology Extension for Medical Domain | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/31ef0507a5c53faae6e36807f0ea6a1442daccc8/13-Table3-1.png)