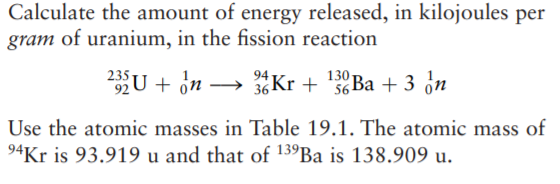Match column I with column II and find the correct answer. Column I Column II (A) Monoploidy (1) 2n - 1 (B) Monosomy (2) 2n + 1 (C) Nullisomy (3) 2n + 2 (D) Trisomy (4) 2n - 2 (E) Tetrasomy (5) n (6) 3n

The energy released per fission of uranium 235 is about 200 MeV. A reactor using U-235 as fuel ... - YouTube
Solved] 5. Draw connections: The uranium fission reactions are triggered by a neutron, and each reaction produces two or three more neutrons. What d... | Course Hero

The ratio of the amounts of energy released as a result of the fusion of 1 kg hydrogen (E(1)) and fission of 1 kg of .(92)U^(236)(E(2)) will be

SOLVED:Calculate the amount of energy released per gram of hydrogen nuclei reacted for the following reaction. The atomic masses are 1^1 H, 1.00782 u; 1^2 H, 2.01410 u ; and an electron,

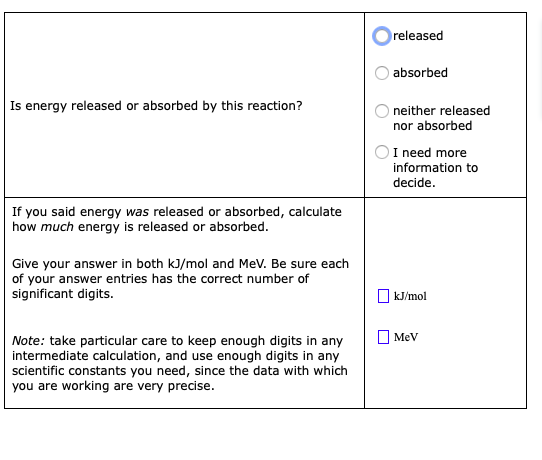
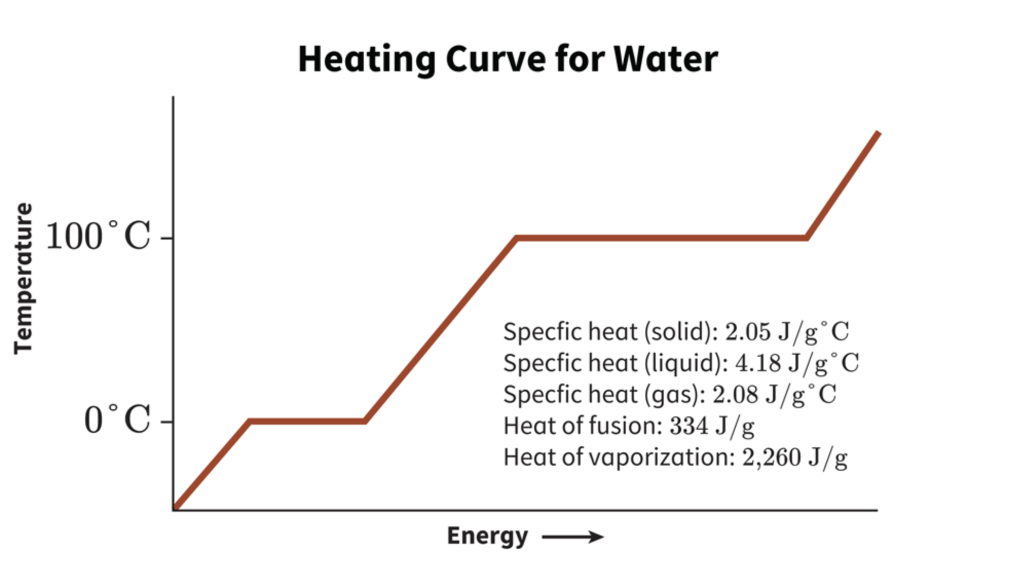
Calculate the energy released in the following nuclear reaction and hence calculate the energy released when 235 gram of uranium-235 undergoes fission. U(92)^(235) + n(0)^(1) to Kr(36)^(92) + Ba(56)^(141) + 3n(0)^(1) Rest

The amount of energy released from a fission reaction is much greater than the energy released from a - Brainly.com
Which is the process in which the energy released per unit mass is more, nuclear fission or fusion? - Quora

The energy released by fission from 2g of ^23592U in kWh is (the energy released per fission is 200MeV ):

The amount of energy released when 1 xx10^10 atoms of chlorine in vapour state are converted to Cl^(–1) ions according to the equation, Cl(g) + e^(-) to Cl^(-) (g) " is "

SOLVED:The energy released during the decay of one (56)/(27) Co atom is 3.72 MeV. If 0.075 M⊙ of cobalt was produced by the decay of 28^56 Ni in SN 1987 A, estimate